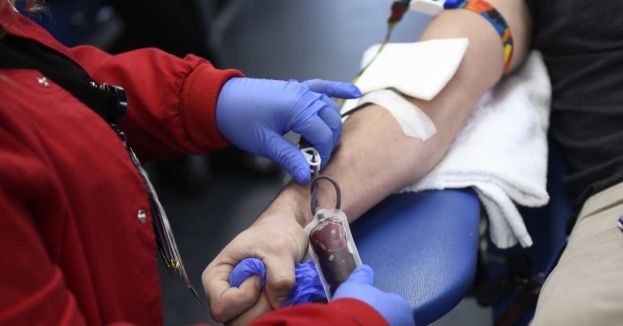
According to the BBC, the Food and Drug Administration (FDA) Commissioner Robert Califf said Friday, “This proposal for an individual risk assessment, regardless of gender or sexual orientation, will enable us to continue using the best science to ensure an adequate and safe blood supply.” Men who had sex with men within three months of giving blood are barred from donating.
With modern technology allowing for screening blood products for infectious diseases, including Human Immunodeficiency Virus (HIV), LGBT advocacy groups have said the current rules on donation were discriminatory. However, in the 1980’’s, lifetime bans were put into place due to the AIDs epidemic. Those bans were later relaxed.
WATCH: 2004 SNL SKETCH: "DONALD TRUMP’S HOUSE OF WINGS”![]()
For several years, a focus on changing federal law on blood donations has been taken up by the LGBT advocacy groups, the American Red Cross, and the American Medical Association (AMA).
January 30, 2023
In December, Tony Morrison from GLAAD shared with NPR that the new changes are what they have been waiting for for many years. He said of the current policy, “It’s a discriminatory policy that assumes that HIV is a gay disease, and it is very much not.”
WATCH: ESPN HOST- DEMOCRAT STRATEGY IS NOT WORKING![]()
In its draft form, the new recommendation indicates that everyone who wants to donate blood will be questioned about whether they’ve had new or multiple sex partners in the past three months and if they’ve engaged in anal sex. If the potential donor answers yes to both questions, they won’t be allowed to donate for three months.
WELP, CLIMATE WARRIORS HAVE THEIR NEXT TARGET PICKED OUT...![]()
HIV-positive people will still be barred from becoming blood donors as they are today to protect the blood supply.
The decision to change the current policy came about to address criticism that it was outdated and discriminatory. Plus, the hope is that more people will donate, which will bolster the nation’s blood bank supplies.
The FDA used a study of 1,600 gay and bisexual men to form its new guidance. They aimed to create screening questions to identify potential donors most likely to test as HIV-positive.
Before publishing the finalized guidelines with changes, the FDA will accept comments from the public for 60 days. The United State’s changes to blood donation guidelines are nothing new. Canada made the change to their 3-month abstinence legislation last year. More recently, Israel, Denmark, Greece, Brazil, France, and the U.K. lifted blood donation restrictions.


 Discover alternative ideas that will make you think
Discover alternative ideas that will make you think Engage in mind bending debate
Engage in mind bending debate Earn points, rise in rank, have fun
Earn points, rise in rank, have fun


